
Huang S, et al. Resolvin E1 sensitizes tumors to immune checkpoint inhibition by stimulating tumor antigen presentation. Cancer Res (2025) 85 (8_Supplement_1): 6912. https://doi.org/10.1158/1538-7445.AM2025-6912

30-40%
response rates
Most cancer patients do not benefit from immune checkpoint inhibitor therapy alone
The Critical Gap Upstream of Checkpoint Blockade
| ▶ |
T-Cell Priming Requirement Immune checkpoint inhibitors release the brakes on the body's natural ability to kill cancer cells, but only when T cells are primed with tumor antigens. |
| ▶ |
Cold Tumor Challenge "Cold" tumors — such as pancreatic, microsatellite stable colorectal, and ovarian — lack primed T cells to respond to checkpoint blockade. |
| ▶ |
Tumor Antigen Expression Requirement Even when T cells are primed by antigen presentation machinery and unleashed by checkpoint blockade, they can clear tumors only if cancer cells express antigens. This requirement must be fulfilled to deliver durable responses. |
TP-317 Activates the Immune Responses Required for Checkpoint Blockade

| ▶ |
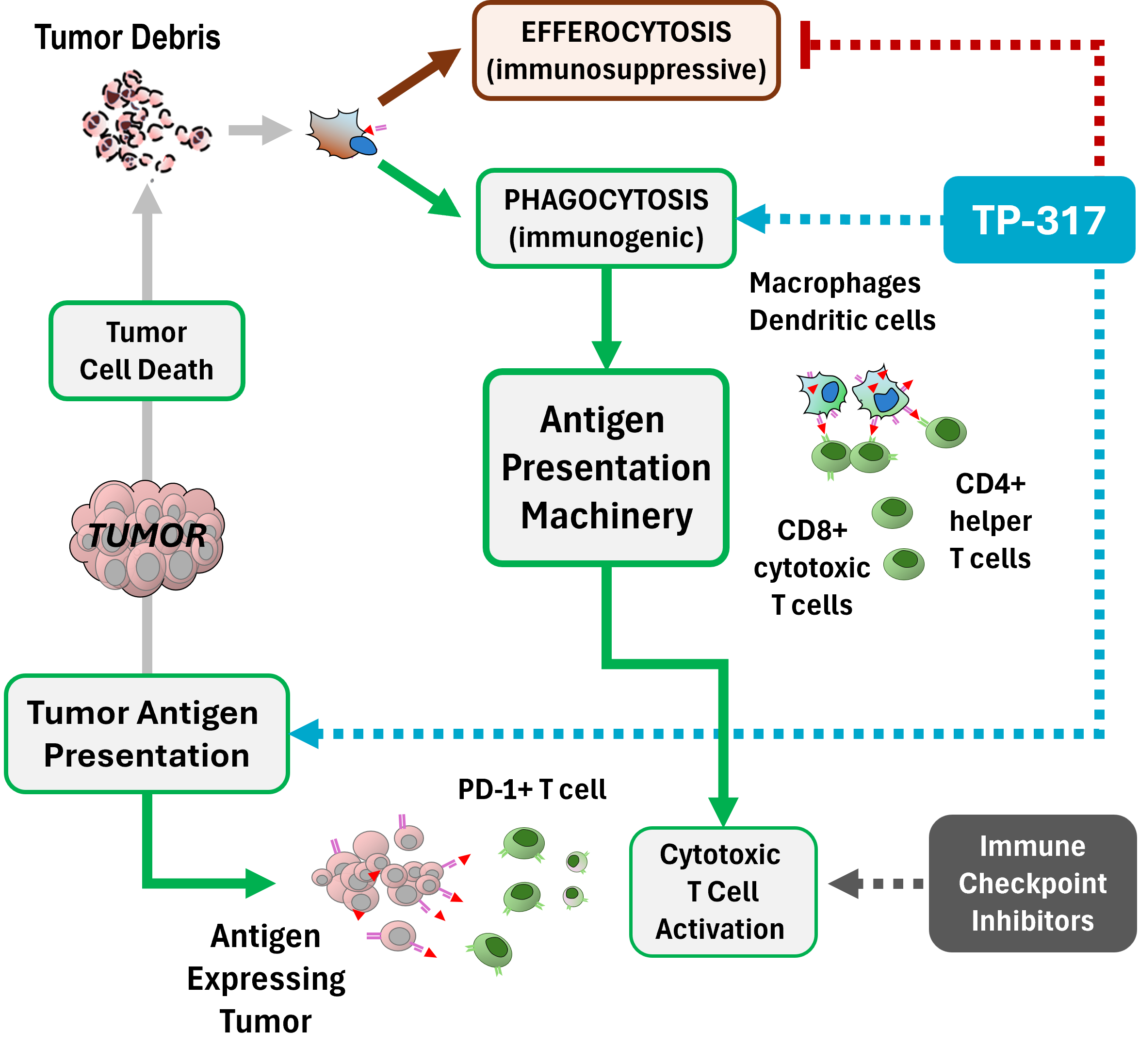
Antigen Presentation TP-317 promotes immunogenic phagocytosis of tumor debris, activating antigen presentation and T-cell priming rather than the immune silencing typical of efferocytosis. |
| ▶ |
Reduced Immune Tolerance By activating immunogenic clearance, TP-317 displaces efferocytosis that causes immune tolerance and tumor progression. |
| ▶ |
Tumor Reprogramming TP-317 reprograms tumors to present antigens — an effect confirmed by single-cell RNA analysis in multiple tumor types. This enables cytotoxic T-cells unleashed by checkpoint blockade to find and kill cancer cells. |
Preclinical Support for Advancement into Cancer Patients
Single Agent Activity
TP-317 has significant single-agent activity, generally on par with dual standard of care therapies (Dual SOC)
Combination Superiority
Adding TP-317 to Dual SOC improves tumor control by more than 50% across syngeneic models.
Immune Pathway Activation
TP-317 upregulates antigen presentation, IFN signaling, and T-cell activation pathways
On-Target Mechanism
Anti-tumor activity is BLT1-dependent, supported by pharmacologic antagonism studies in multiple tumors.
Dual SOC = [anti-PD-1 + chemo] or
[anti-PD-1 + anti-CTLA-4]
Huang S, et al. Resolvin E1 sensitizes tumors to immune checkpoint inhibition by stimulating tumor antigen presentation. Cancer Res (2025) 85 (8_Supplement_1): 6912. https://doi.org/10.1158/1538-7445.AM2025-6912