
References
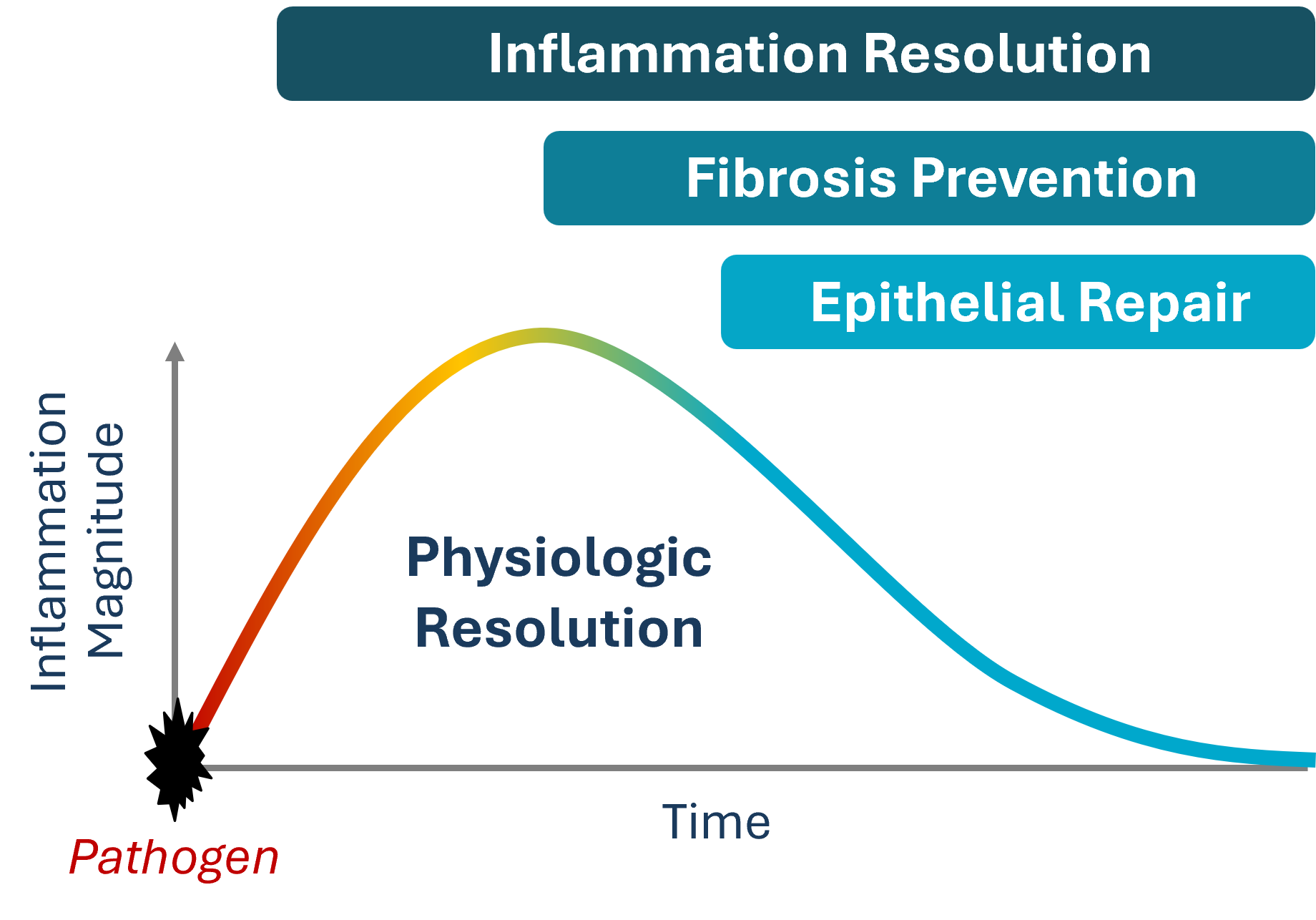
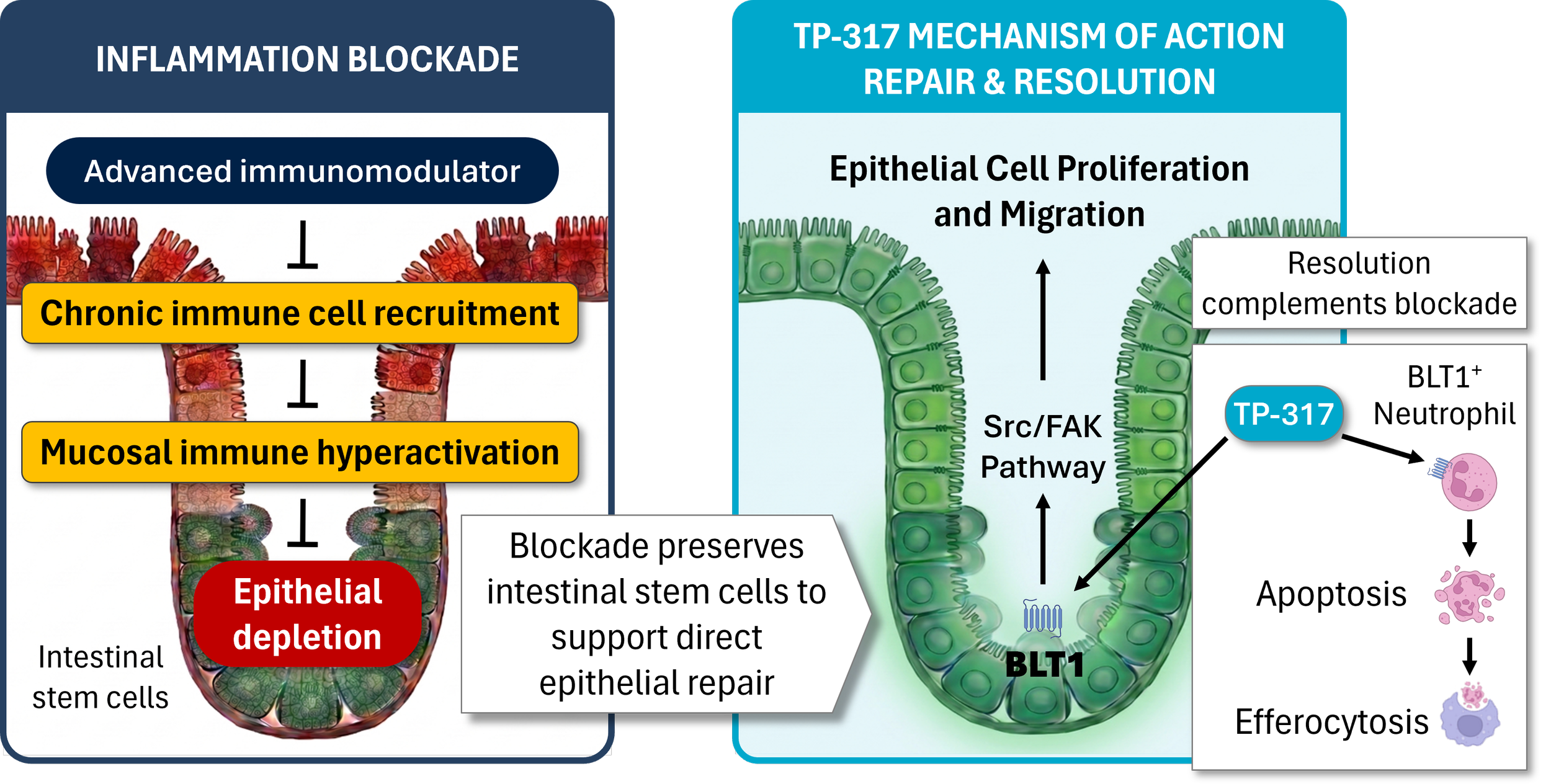
RvE1 Biology
Quirós M, et al. Resolvin E1 is a pro-repair molecule that promotes intestinal epithelial wound healing. Proc Natl Acad Sci U S A. 2020;117(17):9477-9482. https://doi.org/10.1073/pnas.1921335117
Hayashi S, et al. Intestinal epithelial BLT1 promotes mucosal repair. JCI Insight. 2022;7(23):e162392. https://insight.jci.org/articles/view/162392
El Kebir D, Gjorstrup P, Filep JG. Resolvin E1 promotes phagocytosis-induced neutrophil apoptosis and accelerates resolution of pulmonary inflammation. Proc Natl Acad Sci U S A. 2012;109(37):14983-14988. https://doi.org/10.1073/pnas.1206641109
Merlin J, et al. Multipathway In Vitro Pharmacological Characterization of Specialized Proresolving G Protein-Coupled Receptors. Mol Pharmacol. 2022 Apr;101(4):246-256. https://pubmed.ncbi.nlm.nih.gov/35125345/
Arita M, et al. Resolvin E1 selectively interacts with leukotriene B4 receptor BLT1 and ChemR23 to regulate inflammation. J Immunol. 2007;178(6):3912-3917. https://doi.org/10.4049/jimmunol.178.6.3912
Pellas TC, et al. LTB4-induced transient neutropenia in the rat: a model for evaluating efficacy and bioavailability of LTB4 receptor antagonists. J Pharmacol Toxicol Methods. 1993 Nov;30(3). https://doi.org/10.1016/1056-8719(93)90036-e
TP-317 Conference Abstracts
Verstockt B, et al. TP-317, a novel BLT1 agonist oral therapy for inflammatory bowel disease, exhibits anti-inflammatory and epithelial barrier protective efficacy in murine DSS colitis and TNFΔARE Ileitis. Journal of Crohn's and Colitis. 2025;19(Supplement_1). https://doi.org/10.1093/ecco-jcc/jjae190.0243
Brierley S, et al. TP-317, An oral BLT1 agonist, reduces abdominal pain to colorectal distension and reduces key biomarkers of colitis in a mouse model of inflammatory bowel disease. DDW ePoster Library. 05/06/2025; 4157089
Sands B, et al. Pharmacokinetics, pharmacodynamics, safety, and efficacy of oral Resolvin E1-based therapy in inflammatory bowel disease: translating RvE1 activation of BLT1 in experimental models to healthy volunteers. DDW ePoster Library. 05/03/2025; 4154830
Selected IBD References
Danese S, Solitano V, Jairath V, Peyrin-Biroulet L. The future of drug development for inflammatory bowel disease: the need to ACT (advanced combination treatment). Gut. 2022;71(12):2380-2387. https://doi.org/10.1136/gutjnl-2022-327025
Beaugerie L, Kirchgesner J. Balancing Benefit vs Risk of Immunosuppressive Therapy for Individual Patients With Inflammatory Bowel Diseases. Clin Gastroenterol Hepatol. 2019;17(3):370-379. https://doi.org/10.1016/j.cgh.2018.07.013
